UHS to conduct clinical trail of nasal spray to cure COVID-19
Published November 29, 2022
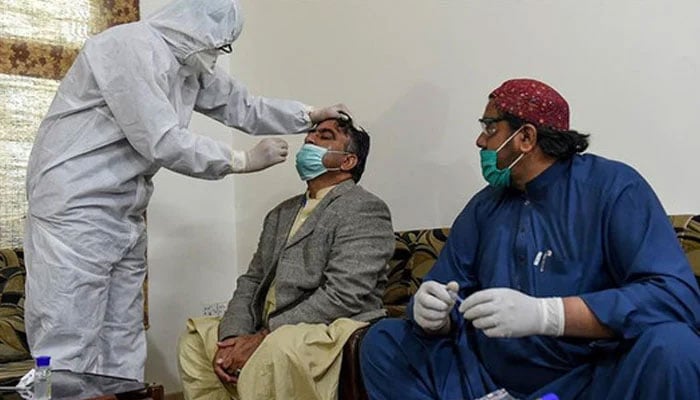
LAHORE: The University of Health Sciences (UHS) is set to conduct a clinical trial of a nasal spray which is meant to cure COVID-19 patients in Pakistan, The News reported Tuesday.
Xlear is a natural sugar-based anti-viral nasal spray which is developed in the United States. If the results of the trial are positive, it has the potential to provide another tool for fighting the COVID-19 virus.
The trial will be completed by January 2023 and is funded and sponsored by Utah-based American Company XLEAR.
Dr Isaac John Founder and CEO of Metanoic Health Limited and Associate Director of Research and Development at Ashford and St Peter’s Hospitals NHS Foundation Trust, Chertsey, Surrey, UK and a clinical researcher from the same institute Dr Meera Nadir coordinated and managed the trial.
In this regard, a training session for University of Health Sciences researchers was held on Monday. Dr John briefed the participants on the protocols of the trial. On this occasion, UHS Vice Chancellor Prof Ahsan Waheed Rathore said that our faculty would benefit from the experience of British experts in research and development.
The trial will test the efficacy of the nasal spray in collaboration of Lahore General Hospital. As many as 80 volunteers will be part of this Phase 2 clinical trial. Prof Rathore and Director Research Professor Saqib Mahmood will lead the research team from Pakistan during the trial.
The intranasal Xlear spray is a natural product extracted from plants. The Xylitol-based anti-viral spray cleanses the respiratory pathway. It moisturises and soothes the sinus and nasal passages while acting like soap for your nose. The spray is available in the market in North America and the European Union countries for more than a decade. It is a very safe anti-viral product but has never been tested for COVID.