COVID-19: AstraZeneca vaccine to be withdrawn due to 'dangerous side effect'
Published May 08, 2024
- AstraZeneca has admitted its vaccine causing TTS, blood clots.
- Company's Vaxzevria has been linked with at least 81 deaths in UK.
- Vaccine manufacturer is being sued by 50 people for its side effects.
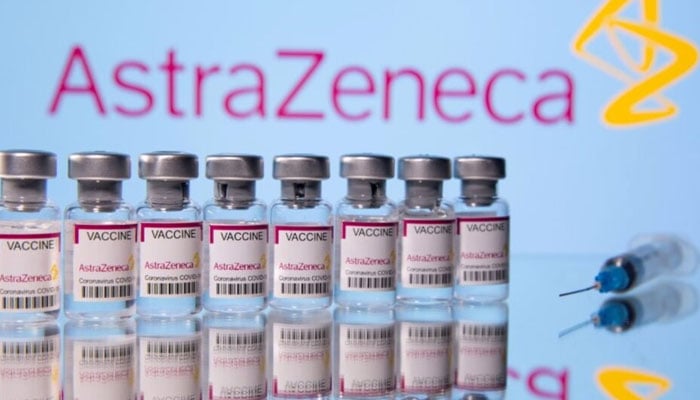
Renowned pharmaceutical company AstraZeneca has announced the withdrawal of its COVID-19 vaccine Vaxzevria globally months after acknowledging vaccine's "rare and dangerous side effect," The Telegraph reported.
As per the publication, the said vaccine has been under scrutiny over a side effect that results in blood clots and low blood platelet counts.
The development comes as the company, which has admitted to its vaccine causing Thrombosis with Thrombocytopenia Syndrome (TTS) on rare occasions, is facing legal action by 50 alleged victims and relatives of deceased individuals in a high court for causing the aforesaid side effect.
So far, at least 81 deaths in UK have been associated with the Vaxzevria vaccine.
However, the company had cited "fall in demand" as the reason behind its decision to withdraw the vaccine.
"As multiple, variant COVID-19 vaccines have since been developed there is a surplus of available updated vaccines," the company said in a statement on Tuesday.
The statement further underscored that the excess stock of newer vaccines has resulted in decreased demand for Vaxzevria, which is no longer being manufactured or supplied.
Furthermore, the manufacturer will also be withdrawing Vaxzevria's marketing authorisations within Europe.
The firm's application to withdraw the vaccine was made on March 5 and came into effect on May 7, according to The Telegraph, which first reported the development.
London-listed AstraZeneca began moving into respiratory syncytial virus vaccines and obesity drugs through several deals last year after a slowdown in growth as COVID-19 medicine sales declined.
According to the company, as quoted by The Guardian, more than 6.5 million lives were saved in the first year of use alone and over 3 billion doses were supplied globally.
AstraZeneca's COVID-19 vaccine was also approved for use by the Pakistani government in its bid to curb the rapid spread of the pandemic in the country.
The said vaccine comprises of another virus which is essentially modified to have the gene for making a protein from coronavirus causing SARS-CoV-2.