MHRA issues recall of popular antidepressant after patient discovers wrong drug
Published April 29, 2026
The UK health watchdog MHRA issued a recall for a commonly prescribed antidepressant across the UK.
The recall comes after a patient found the wrong drug inside their sealed packaging.
More than 80,000 packs of a popular antidepressant were recalled by the UK Medicine and Healthcare Products Regulatory Agency on Wednesday, April 28, 2026.
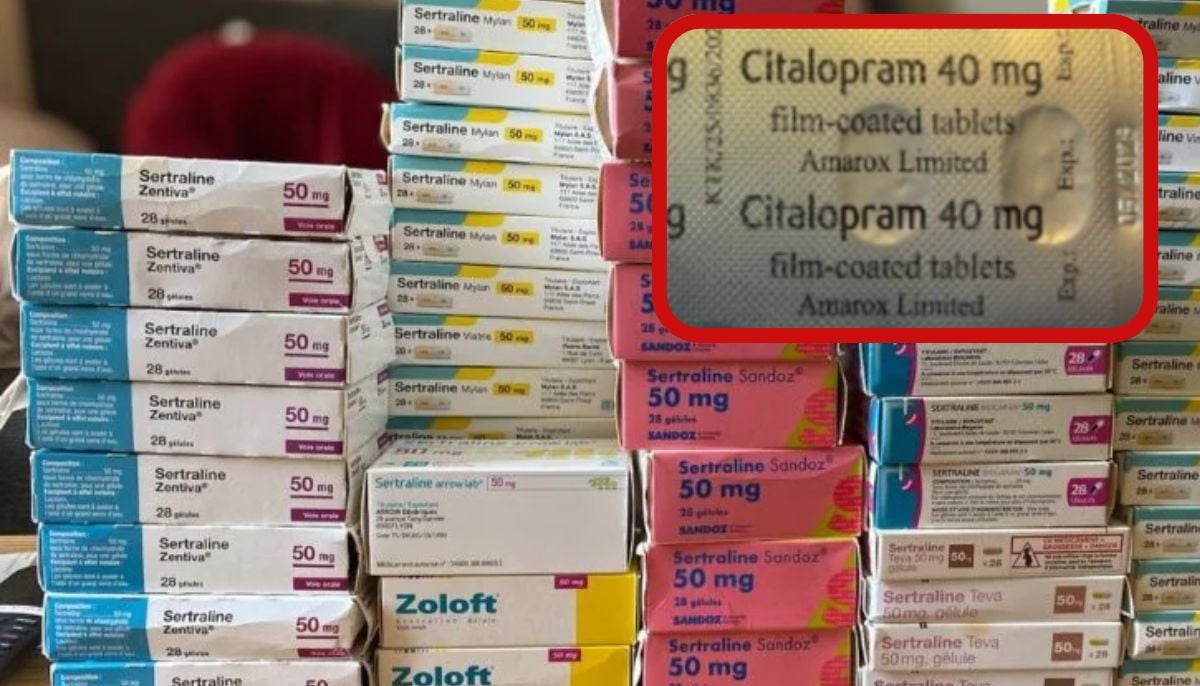
The patient, who was prescribed Sertraline 100mg film-coated tablets, discovered a packet of Citalopram 40mg film-coated tablets, another antidepressant, inside the carton.
According to the MHRA statement, following the reports, UK-based pharmaceutical Amarox issued a “precautionary recall” of a specific batch of sertraline.
The UK health regulatory agency further said that “both medications are produced by the same site.
The error appears to have occurred during secondary packaging of the blister strips into the cartons.”
Patients who believe they have already taken any citalopram 40mg tablets by mistake, or are experiencing side effects should seek medical advice immediately.
The UK health agency further warned that pharmacists should stop supplying the batch and contact any patients who may have received the affected product.
For the unversed, commonly used antidepressants like sertraline and citalopram are both selective serotonin reuptake inhibitors, or SSRIs.
These antidepressants are used to treat depression, anxiety disorders, and other related mental health issues by raising levels of serotonin, which is a key brain chemical.